 
The overall survival benefit was also observed in patients with a PD-L1 expression level of less than 1%, with a median duration of 17.2 months (95% CI, 12.8 to 22.0) with nivolumab plus ipilimumab and 12.2 months (95% CI, 9.2 to 14.3) with chemotherapy. The median duration of response was 23.2 months with nivolumab plus ipilimumab and 6.2 months with chemotherapy. The primary end point reported here was overall survival with nivolumab plus ipilimumab as compared with chemotherapy in patients with a PD-L1 expression level of 1% or more.Īmong the patients with a PD-L1 expression level of 1% or more, the median duration of overall survival was 17.1 months (95% confidence interval, 15.0 to 20.1) with nivolumab plus ipilimumab and 14.9 months (95% CI, 12.7 to 16.7) with chemotherapy (P = 0.007), with 2-year overall survival rates of 40.0% and 32.8%, respectively. All the patients had received no previous chemotherapy. The patients who had a PD-L1 expression level of less than 1% were randomly assigned in a 1:1:1 ratio to receive nivolumab plus ipilimumab, nivolumab plus chemotherapy, or chemotherapy alone. In this open-label, phase 3 trial, we randomly assigned patients with stage IV or recurrent NSCLC and a PD-L1 expression level of 1% or more in a 1:1:1 ratio to receive nivolumab plus ipilimumab, nivolumab alone, or chemotherapy.  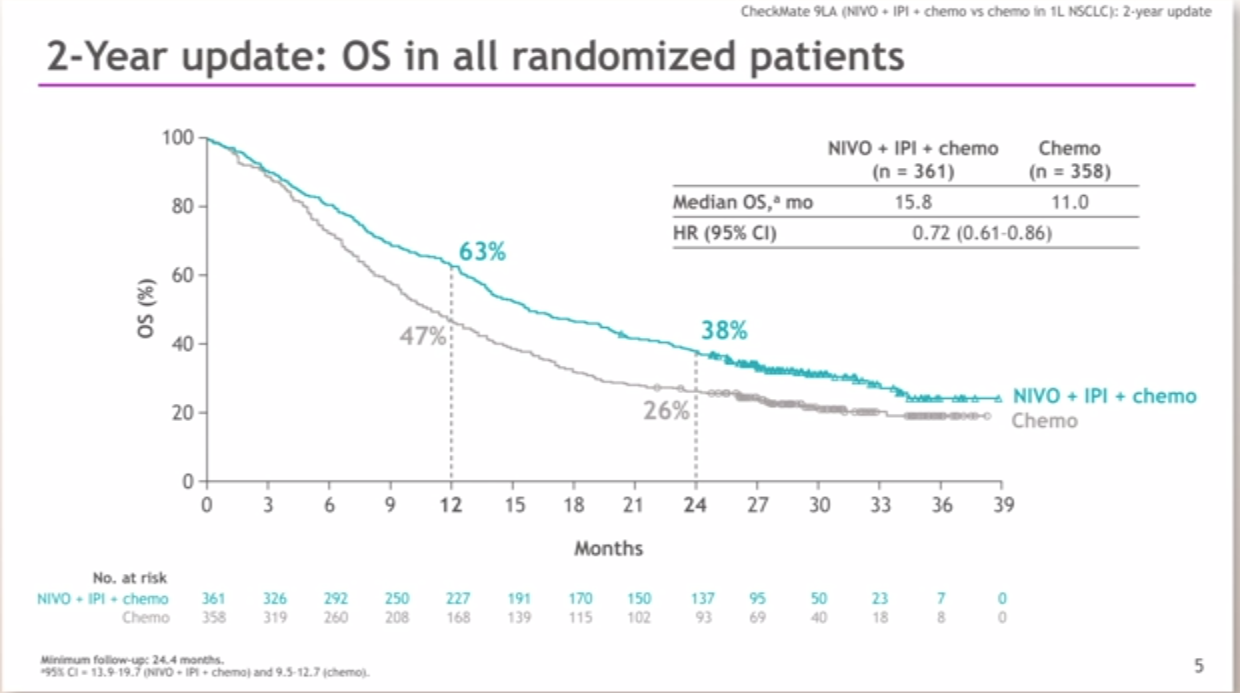
Data are needed to assess the long-term benefit of nivolumab plus ipilimumab in patients with NSCLC. In an early-phase study involving patients with advanced non-small-cell lung cancer (NSCLC), the response rate was better with nivolumab plus ipilimumab than with nivolumab monotherapy, particularly among patients with tumors that expressed programmed death ligand 1 (PD-L1). Alexandru Trestioreanu, Bucharest, Romania (A.A.) the Hospital Italiano de Buenos Aires, Buenos Aires (L.L.) Instituto Jalisciense de Cancerologia, Guadalajara, Mexico (E.M.J.) the Saitama Cancer Center, Saitama, Japan (H.S.) Matrai Gyogyintezet, Matrahaza, Hungary (I.A.) Limoges University Hospital, Limoges (A.V.), and Aix-Marseille University, National Center for Scientific Research, INSERM, Centre de Recherche en Cancérologie de Marseille, Assistance Publique-Hôpitaux de Marseille, Marseille (F.B.) - all in France Centre Hospitalier Universitaire Vaudois, Lausanne University, Lausanne, Switzerland (S.P.) Sotiria General Hospital, National and Kapodistrian University of Athens, Athens (K.S.) Lung Clinic Grosshansdorf, Airway Research Center North, German Center of Lung Research, Grosshansdorf, Germany (M.R.) Fox Chase Cancer Center, Philadelphia (H.B.) Johns Hopkins Kimmel Cancer Center, Baltimore (J.R.B.) Princess Alexandra Hospital, Brisbane, QLD, Australia (K.J.O.) Bristol-Myers Squibb, Princeton, NJ (W.J.G., P.B., S.K.R., R.S.K., F.E.N.) and Winship Cancer Institute, Emory University, Atlanta (S.S.R.). 1 From the Memorial Sloan Kettering Cancer Center, New York (M.D.H.) Hospital Universitario Doce de Octubre, Centro Nacional de Investigaciones Oncológicas, Universidad Complutense, and Centro de Investigación Biomédica en Red de Cáncer, Madrid (L.P.-A.), Hospital Universitario Virgen Del Rocio, Seville (R.B.C.), and the Catalan Institute of Oncology-Germans Trias i Pujol Hospital, Badalona (E.C.C.) - all in Spain Ambulatorium Chemioterapii, Bydgoszcz, Poland (B.Z.) the Asan Medical Center (S.-W.K.) and the Samsung Medical Center at Sungkyunkwan University School of Medicine (K.P.) - both in Seoul, South Korea the Institute of Oncology Prof.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
